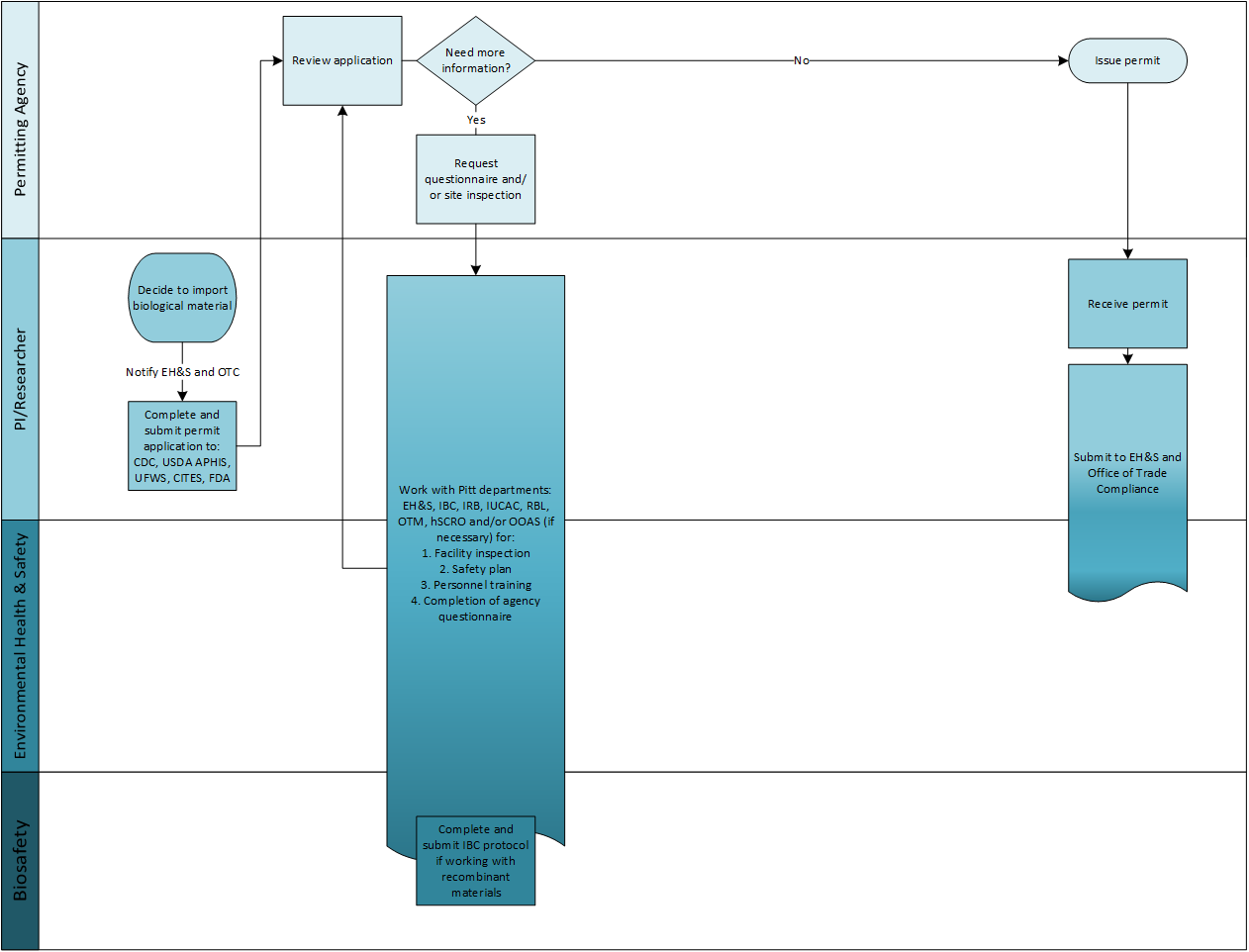
Depending on the materials and techniques being used in your experimental protocols - you may need approval from various University of Pittsburgh departmental committees:
- Environmental Health & Safety (EH&S)
- Institutional Biosafety Committee (IBC)
- Institutional Animal Care and Use Committee (IACUC)
- Institutional Review Board (IRB)
- Human Stem Cell Research Oversight (hSCRO)
- Office for Oversight of Anatomical Specimens (OOAS)
- Division of Laboratory Animal Resources (DLAR)
- Radiation Safety Office
- Regional Biocontainment Laboratory (RBL)
- Office of Innovation and Entrepreneurship
- Export Control
- Conflict of Interest office (COI)
These departments will also need to review and approve your material transfer agreement in the MyRA system.
Please visit https://www.osp.pitt.edu/ccc-material-transfer-agreements for more information on material transfer agreements!
